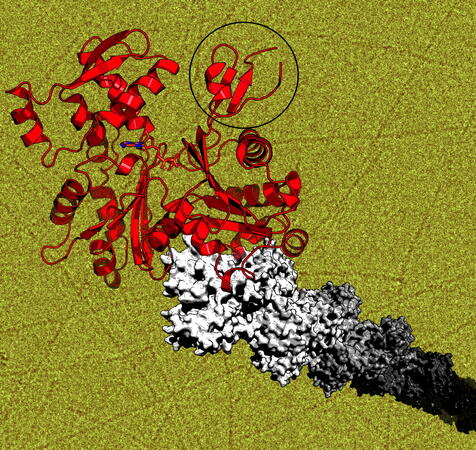
Molecular mechanisms of parasite motility
Malaria is one of the world's most devastating infectious diseases. Each year, nearly half a million people die of malaria. The disease is caused by Plasmodium spp., which comprise a group of unicellular, eukaryotic, intracellular parasites, belonging to the phylum Apicomplexa. They use an actomyosin motor complex, termed glideosome, for rapid gliding motility and host cell invasion. The motor components are to a large extent unique to these parasites or highly diverged from the corresponding human proteins. Parasite actin filaments are short, and their rapid treadmilling is regulated by an unusually small number of actin-binding proteins.
Our work is focused on understanding malaria parasite gliding motility and the molecular machinery behind at the molecular/atomic level. A broad range of structural biology and biochemical/biophysical methods is used for this aim.
Our work is aimed at a mechanistic understanding of gliding motility, which malaria parasites and their relatives use for getting to and invading their host cells. We employ a broad range of biochemical, biophysical and hybrid structural biology methods for creating a complete molecular picture of the parasite actin-myosin motor and the entire glideosome. We also want to understand the evolution of apicomplexan gliding motility and eukaryotic actin-myosin motors in a broader sense. Understanding the mechanistic differences in cell motility between parasites and humans may, furthermore, open up new avenues for treatment and/or prevention of malaria.
We have determined atomic structures of malaria parasite actins in both monomeric and filamentous form and characterized their polymerization properties as well as nearly all the parasite actin-binding proteins known to date. Our future work is directed more towards larger complexes and reconstructing the entire parasite glideosome for structural and functional characterization.
My research group is divided between the Faculty of Biochemistry and Molecular Medicine at the University of Oulu, Finland, and the Department of Biomedicine, University of Bergen, Norway.




